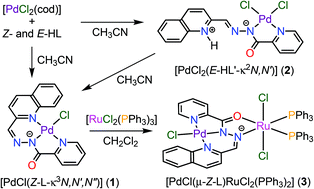
Palladium(ii) mononuclear and palladium(ii)/ruthenium(ii) heterodinuclear complexes containing 2-quinolyl-substituted (pyridine-2-carbonyl)hydrazone - Dalton Transactions (RSC Publishing)

Reduction of α,β-unsaturated carbonyl compounds by palladium(II) and nickel(II) complexes having nitrogen-containing ligands - ScienceDirect

Palladium‐Catalyzed Selective Reduction of Carbonyl Compounds - Sarkar - 2020 - European Journal of Inorganic Chemistry - Wiley Online Library

Low Coordinate Germanium(II) and Tin(II) Hydride Complexes: Efficient Catalysts for the Hydroboration of Carbonyl Compounds | Chem-Station Int. Ed.
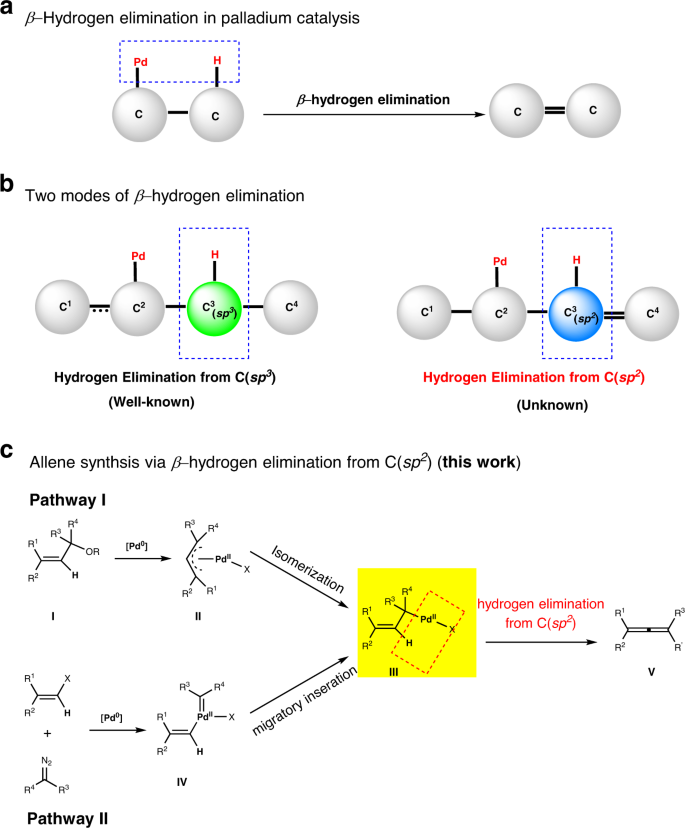
Palladium-catalyzed allene synthesis enabled by β-hydrogen elimination from sp2-carbon | Nature Communications

Umpolung of Carbonyl Groups as Alkyl Organometallic Reagent Surrogates for Palladium‐Catalyzed Allylic Alkylation - Zhu - 2018 - Angewandte Chemie - Wiley Online Library
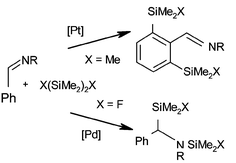
Palladium or platinum complex catalysed reactions of carbonyl and imine compounds with disilanes - Dalton Transactions (RSC Publishing)

Asymmetric carbonyl-ene reaction of trifluoropyruvate catalyzed by Pd(II)-SunPhos complex - ScienceDirect

Palladium nanoparticles on amino-modified silica-catalyzed C–C bond formation with carbonyl insertion | SpringerLink
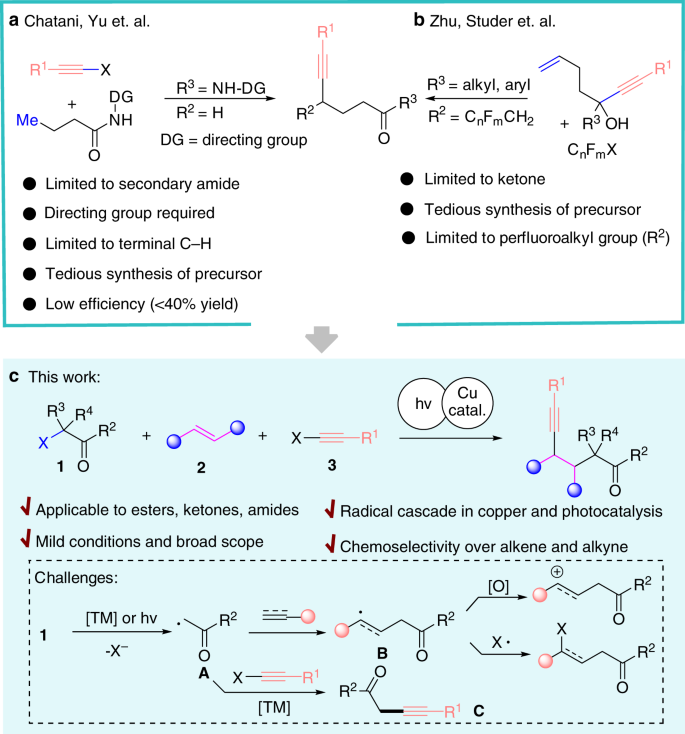
Unified and practical access to ɤ-alkynylated carbonyl derivatives via streamlined assembly at room temperature | Communications Chemistry
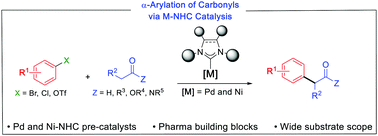
N-Heterocyclic carbene complexes enabling the α-arylation of carbonyl compounds - Chemical Communications (RSC Publishing)

Diastereoselective Carbonyl Allylation with Simple Olefins Enabled by Palladium Complex-Catalyzed C-H Oxidative Borylation
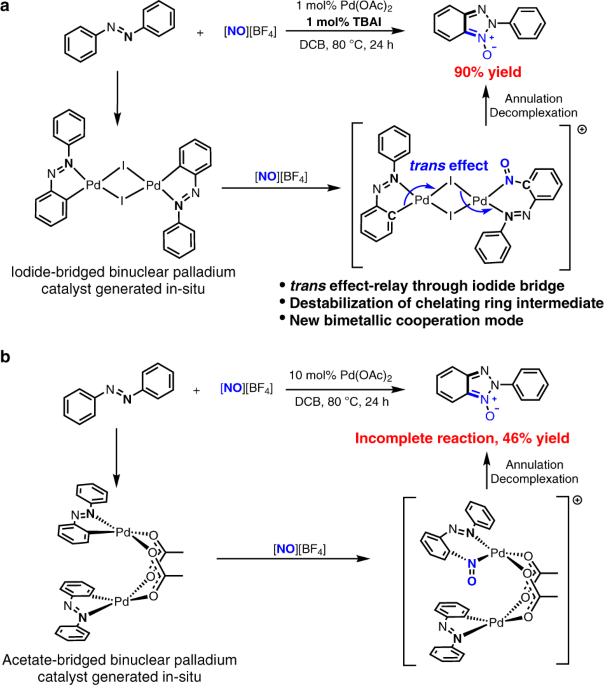
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry

Molecules | Free Full-Text | Synthesis, Characterization, and Reaction Studies of Pd(II) Tripeptide Complexes | HTML

One‐Pot Syntheses of α,β‐Unsaturated Carbonyl Compounds through Palladium‐Mediated Dehydrogenation of Ketones, Aldehydes, Esters, Lactones and Amides - Muzart - 2010 - European Journal of Organic Chemistry - Wiley Online Library

Bulky N‐Heterocyclic‐Carbene‐Coordinated Palladium Catalysts for 1,2‐Addition of Arylboron Compounds to Carbonyl Compounds - Okuda - 2020 - ChemCatChem - Wiley Online Library
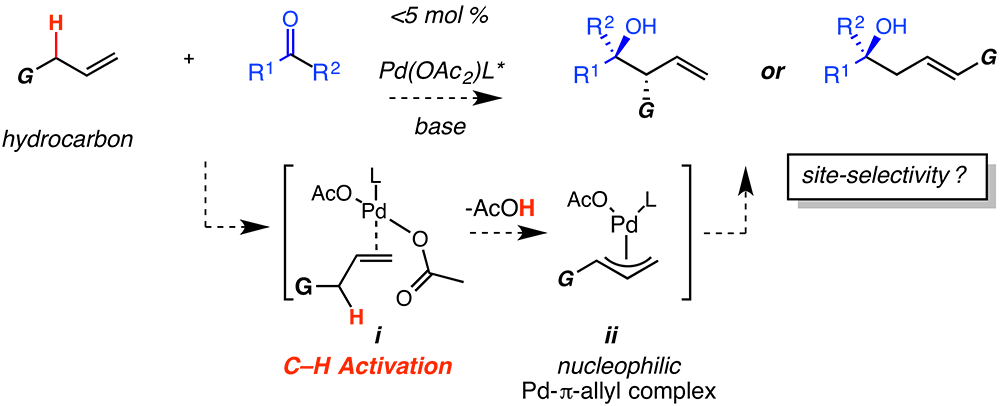
Report: Metal Catalyzed C-H Bond Activation and Allyl Addition: Development of New Transition Metal Catalysis for Practical and Uniquely Efficient Carbon-Carbon Bond Forming Reactions (58th Annual Report on Research Under Sponsorship of

![20. M(0) carbonyl complexes with hybrid NHC ligands [114,120-122]. | Download Scientific Diagram 20. M(0) carbonyl complexes with hybrid NHC ligands [114,120-122]. | Download Scientific Diagram](https://www.researchgate.net/profile/Lu-Jiang-5/publication/272102102/figure/fig8/AS:572844641271822@1513588213512/M0-carbonyl-complexes-with-hybrid-NHC-ligands-114-120-122_Q640.jpg)



